Fabrication of Biological Materials Subcellular Dosing
Fabrication of Microstructured Biological Materials Using Multiphoton Excitation
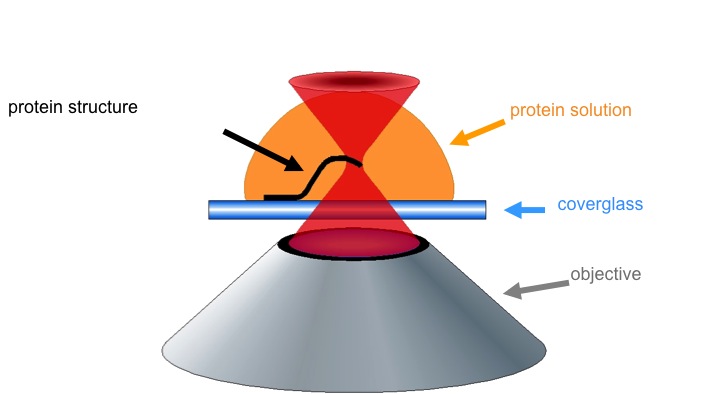 The Shear Lab uses multiphoton excitation (MPE) to perform site-specific photochemistry such as fabrication of 3D materials. A titanium:sapphire laser beam focused through a microscope objective leads to electronic excitation of a photoinitiator/ photosensitizer in a protein solution. The high intensity required restricts reactions to the focal volume. Localized excited-state processes create small-dimension solids that can be directly written into microstructures by moving the microscope stage (coverglass).
The Shear Lab uses multiphoton excitation (MPE) to perform site-specific photochemistry such as fabrication of 3D materials. A titanium:sapphire laser beam focused through a microscope objective leads to electronic excitation of a photoinitiator/ photosensitizer in a protein solution. The high intensity required restricts reactions to the focal volume. Localized excited-state processes create small-dimension solids that can be directly written into microstructures by moving the microscope stage (coverglass).
Dynamic Mask-based Multiphoton Lithography
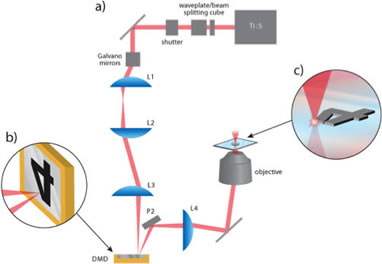
We demonstrate a significant advancement in the optical technology used to pattern 3D microstructures. Our strategy uses a digital micromirror device (DMD) to control the spatial irradiation pattern of a scanning laser beam focused by a high numerical aperture objective. The graphic image on the DMD is reproduced by high intensity laser light at the focal plane resulting in spatially resolved photochemical events that lead to the deposition of solid micron-scale structures.
Above: Schematic of the DMD-directed multiphoton lithography instrument. (a) Output from pulsed laser is attenuated by waveplate and beamsplitting cube, a galvo mirror scanner translates the beam in a raster pattern and three relay lenses direct the beam onto the face of the DMD. (b) Depiction of focused beam scanning over the DMD chip. (c) Illustration of the image on the DMD being replicated by nonlinear laser microfabrication.
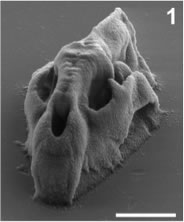

Above: High resolution X-ray CT data from digimorph.org were used to fabricate a series of micro-reconstructions of biological organisms from a precursor solution of bovine serum albumin (BSA). The synchronization of DMD image sequences with vertical sample plane steps resulted in 3D structures. The microstructures consisted of 60-120 individually defined planes. Each took plane took ~1 to 2 seconds to fabricate. Scale bars, 10 μm
Bacterial Group Behavior
Dynamic mask-directed multiphoton lithography allows for rapid prototyping of chemically responsive micro-architectures suitable to grow bacterial colonies to study group behaviors in controlled environments. Bacteria are social organisms and engage in a process termed quorum sensing (QS). Cells use small molecules to communicate within a population and coordinate gene expression to accomplish tasks that would otherwise be difficult for an individual cell. Protein structures permit sufficient transport of waste and nutrients to grow isolated high-density/low-cell-number bacterial colonies. Using multiphoton-fabricated microstructures, we have the ability to study QS-dependent behaviors in Pseudomonas aeruginosa are modulated by certain variables (cell number, cell density, and flow rate through the system). Additionally, we have shown that P. aeruginosa cells develop antibiotic resistance as cell density increases within as few as ~150 cells.
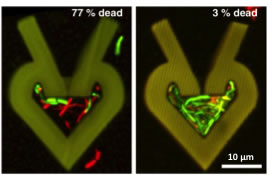
Above: Confocal reconstructions of protein microstructures dosed for 2 h with gentamicin at the minimum inhibitory concentration (MIC) demonstrate that 77% of cells growing at a low density are dead after antibiotic treatment (left). In comparison, only 3% of cells growing at a higher density (right) are susceptible to the gentamicin. Dead and live cells appear red and green, respectively.
Tunable Properties of Fabricated Microstructures

The design of responsive, tunable, bio-compatible smart materials would be of great value for the advancement of microfluidics, biosensors, tissue scaffolds, and single cell analysis. Combining tunable properties with the high-resolution and three-dimensional physical features inherent to multiphoton excitation allows for greater control and manipulation of analytical systems. In recent studies, we have developed tunable multiphoton-fabricated protein hydrogels with high-resolution (i.e., low- to sub-micrometer) three-dimensional topographical features that respond to changes in their local chemical (pH, ionic strength…) and physical (temperature) environment in definable ways. When exposed to elevated temperatures, the materials undergo irreversible volume changes, where the size change of a protein hydrogel is dictated by its photofabrication conditions. Microstructures composed of various proteins, including bovine serum albumin (BSA), lysozyme, avidin, and casein have been investigated and have shown to respond to increasing temperatures and rapid pH changes in varying ways. Of these proteins, for example, BSA hydrogels undergo a volume increase with increasing temperature, while avidin hydrogels undergo a volume decrease. The development of tunable, biocompatible hydrogels has been applied to the study of single cells, particularly for the study of isolated bacterium (above).
DMD-based Fabrication with Acrylate Resin

Acrylate resin has been used to create structures with an inexpesive Q-switched Nd:YAG laser. Although acrylate is not a biomaterial, such structures could be used as master structures to make molds. The molds could then be used to make identical structures from a range of polymers and biopolymers.
3D Microfabrication in Cell Culture
This project focuses on optimizing hyaluronic acid (HA) hydrogels as scaffold materials for neuronal cells. HA-based hydrogels are modified with internal, 3D-patterned protein structures through a photochemical 'direct-write' process based on multiphoton excitation, where photocrosslinking is confined to the focal volume of a laser. Additionally, we are investigating the interaction of cells with HA hydrogels by a parallel approach, which aims to elicit any effects of hydrogel mechanical properties on neuronal and glial phenotypes in 3D culture.


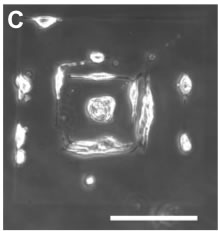
Above: 3D MPE crosslinked BSA structures within hydrogels. A) Representation of mask used to fabricate helices. B) 3D reconstructions of confocal microscope images of BSA-FITC helices fabricated inside an HA hydrogel. The gridded boxes have side lengths of 375 µm and a depth of 36 µm. C) Dissociated dorsal root ganglia on biotinylated BSA structures modified with biotin-IKVAV-peptides via neutravidin. Structures were fabricated in a pattern of concentric squares (left image; outer square side length = 200 µm, each inner square decreased in side length by 2 µm, total of 49 squares). Scale bar = 100 µm.
Mechanical Properties of Fabricated Microstructures

The mechanical properties of cell substrates affect cell differentiation, growth and motility. Thus the mechanical properties of a material are important to consider when designing a cell scaffold. In the image on the right, single strands of bovine serum albumin (BSA) matrix have been fabricated across a trench cut in a glass slide. Using an atomic force microscope (AFM), the mechanical properties of these matrix strands can be measured. Preliminary data suggests that the Young's modulus of protein matrix can be modified by varying fabrication parameters.
